Food Temperature FAQ
Because the temperature of the food can be different in different places at the same time. It is not unusual for the internal temperature of a large roast or turkey to vary by as much as 20 to 30°F (10 to 15°C) throughout the meat or bird. Even a steak or a boneless chicken breast may show differences of many degrees as you move the tip of your thermometer probe from the surface toward the center of the piece, or from end to end, depending upon the speed and accuracy of your thermometer.
Please keep in mind that meat will continue to cook after you take it off the heat. This is called "resting." Cooked meat should be allowed to "rest" after cooking and before cutting. This permits the juices to be reabsorbed into the fibers of the meat. If you skip resting, you will lose more juices when the meat is cut. The temperature of the meat will always continue to rise a little during the resting period. Typically, even a small steak or individually cooked piece of chicken will rise at least three or four degrees during resting. A larger roast or turkey can rise as much as ten to fifteen degrees depending upon conditions. So you should remove your meat from the oven or grill prior to reaching your target temperature. Otherwise, it will be overcooked. Read the ultimate guide to resting meat here.
Never leave a thermometer inside an oven, grill, smoker, or microwave while cooking unless it is specifically designed for this. Don't leave your thermometer on the grill hood or close to an open flame. Some probes are designed to be left inside an oven or grill, but the thermometer housing itself (where the electronic components are kept) typically needs to be kept cool and should only be brought near heat for short periods of time. When checking temperatures above a grill or fire, be careful that the thermometer body does not get too hot.
Because the bone marrow in chicken bones can release blood while cooking. If the chicken has reached at least 71°C for five minutes or more, it is safely done.
The UK Food Agency publishes temperature guidelines for food holding and cooking . You can use your ETI thermometer to check temperatures and minimize food-borne illness in your kitchen. Bacteria thrive between the temperatures of 4.5°C and 60°C. Food should not be stored between these temperatures for extended periods of time. Some leftover foods must be reheated to minimum temperatures to assure sufficient "kill rates" of bacteria or parasites.
Holding hot foods 63°C or higher
Holding cold foods less than 8°C
Fridge temperature 4.5°C or colder
Freezer temperature 18°C to -23°C
See also our chart of chef-recommended food temperatures.
The best way to take an accurate reading of liquids is firstly to stir the liquid. Follow this by stirring the thermometer probe through the liquid, watching the temperature change as it moves through.
Thermometry FAQ
It may seem obvious, but a thermometer is merely a tool that gives you information about the temperature of the food you are preparing. YOU have to make the decisions about when to increase or decrease the heat and about when things are cooked based on the information the thermometer gives. In most cases, you are looking for the highest temperature reached in the thickest part of the food to assess if the food is cooked.
While different people like their meat or other food cooked to different levels of 'doneness', here is a chart of chef-recommended temperatures to get you started.
When testing if your meat is cooked, the coldest part will be the very center of the thickest portion. With larger foods, you can take quick readings with your thermometer in several locations to verify that the entire portion is done. If you are chilling a food, the center of the thickest part will be the last to cool.
Keep in mind that different types of thermometers have sensors of different sizes. A dial thermometer can have a sensor as big as an inch long and your reading will be an average of the temperature of all the different materials touching that sensor. Most digital thermometers have small sensors at their tip. Penetrate the food you are checking with the probe and place the very tip of the probe where you want to measure.
When testing if your meat is cooked, the coldest part will be the very center of the thickest portion. With larger foods, you can take quick readings with your thermometer in several locations to verify that the entire portion is done. If you are chilling a food, the center of the thickest part will be the last to cool.
Keep in mind that different types of thermometers have sensors of different sizes. A dial thermometer can have a sensor as big as an inch long and your reading will be an average of the temperature of all the different materials touching that sensor. Most digital thermometers have small sensors at their tip. Penetrate the food you are checking with the probe and place the very tip of the probe where you want to measure.
The word "calibrate" is often thought to mean making some kind of adjustment to a thermometer for accuracy. While adjustments are sometimes needed, "calibrating" a thermometer simply means testing its accuracy against a verifiable standard. If the thermometer is within the accuracy specification listed by the manufacturer or quality control agency, no adjustment should be made.
When it comes to recommended frequency, there is a very wide range. Mechanical thermometers like dial thermometers should be calibrated very regularly if not daily, while digital thermometers are often only calibrated once a year. Check the stated recommendation for your type and model of thermometer or check with the appropriate health department or quality control agency with authority over your operation.
Primarily, because the temperature of food keeps changing while it is cooking, and your ETI digital thermometer is accurate enough to see it. Thermometers will only "lock in" on a given reading if they have a "hold" feature designed to do that.
One common added feature for electronic thermometers, particularly electronic cooking thermometers, is a splashproof or waterproof rating.
The International Electrotechnical Commission (IEC) has created a standard for rating instruments based on their ability to protect their electronic components from corrosion by water or dust. The International Protection Rating code (or IP code) consists of the letters "IP" followed by two numbers.
The first number tells you how protected an instrument's electronics are from penetration by solids (like dust) and the second number tells you how they stand up against liquids. See the table here.
If, for example, a thermometer had a rating of "IP65," that would mean that it was tested and found completely protected against dust as well as protected against low pressure jets of liquid from all sides, but NOT protected against immersion, or an accidental drop into the soup.
Probably, because the temperature is higher than the ice point unless you take the time to create a properly made ice bath.
If the thermometer is, in fact, outside of its published accuracy specification, adjust accordingly or contact our Service Department: service@etiltd.com, 01903 202151.
Check the battery compartment. In many thermometers there is a small metal clip that holds the batteries in place. Even if the batteries are in place, if they are not under this little clip, the thermometer won't turn on. If the clip is in place and the thermometer still won't turn on, contact the ETI Service Department for advice (service@etiltd.com, 01903 202151).
Most probably, because your old thermometer is not as accurate as your new ETI digital thermometer. Test both thermometers in a properly made ice bath to check and adjust accordingly or contact our Service Department: service@etiltd.com, 01903 202151.
You should wipe the probe clean with cleaning wipes or soap any time it comes in contact with raw meat, and you should wipe the whole thermometer casing after each use being careful not to get it wet. Never put the housing of your digital thermometer near water unless it has an IP rating of 66 or higher. WARNING - IPA and other solvents may cause damage to the case and screen of this instrument.
The boiling point of water is directly dependent on (atmospheric) pressure. It is a physical related property. Water boils at 100°C ONLY if it is pure (de-mineralized) and under atmospheric pressure of 1013.25 hPa.
So at high altitudes or bad weather-low pressure, you may think that the instrument reads the wrong temperature.
For example, Low air pressure at 990 hPa water will boil at 99.3 °C and at higher air pressure 1040 hPa water boils at 100.7 °C.
Infrared FAQ
Infrared thermometers are very fast, typically giving a reading in a fraction of a second, or the time it takes for the thermometer's processor to perform its calculations.
Their speed and relative ease of use have made infrared thermometers invaluable public safety tools in the food service industry, manufacturing, HVAC, asphalt & concrete, labs and countless other industrial applications.
Infrared thermometers are ideal for taking surface temperature measurements from a distance. They provide relatively accurate temperatures without ever having to touch the object you're measuring.
This is can be useful when it's impractical to insert a probe into the item being measured, or if the surface is just out of reach and a surface probe will not do the job. You might use an infrared thermometer to measure objects that are:
Fragile (computer circuitry)
Dangerous (gears, molten metal)
Impenetrable (frozen foods)
Liable to contamination (foods, saline solution)
Moving (conveyor belt, living organisms)
Out of reach (air conditioning ducts, ear drums)
All Infrared thermometers sold by ETI Ltd, that have Laser Pointers, comply with BS EN 60825-1: 2014: - Safety of laser products:
Part 1. Equipment classification and requirements: - Class 2. The basis of this EU Harmonised Standard is that the laser should be strong enough to make someone blink but not strong enough to damage tissue.
This therefore prevents damage to the eye unless someone deliberately stares at the laser light for a long time, causing self-inflicted injury. i.e. For consumer laser products in general there is a requirement that, by employing technical means (i.e. engineering controls), lasers shall not cause damage to the eyes or skin under normal and reasonably foreseeable conditions of use, including momentary accidental or unintended exposure.
Also any laser should not be shone in anyone’s eyes; it can be very distracting and could result in a serious incident.We strongly recommend that the laser is only used to help target the area being measured, indicating the optical ratio of the instrument.
That will depend on the model or type of your infrared thermometer. Read the user manual that came with your infrared thermometer to lear about the full range of features it offers and how to use them.
For accuracy, an infrared thermometer should be kept free of dirt, dust, moisture, fog, smoke and debris. Always take the time to clean your infrared thermometer after exposure to a dirty, dusty, smokey or humid environment. You should also plan a regular cleaning every six months or so. Particular care should be taken to keep the infrared lens or opening clean and free of debris.
To clean your infrared thermometer:
1. Use a soft cloth or cotton swab with water or medical alcohol.
2. Carefully wipe first the lens and then the body of the thermometer.
3. Allow the lens to dry fully before using the thermometer.
4.Never submerge any part of the thermometer in water.
See the video of infrared basics here.
Infrared thermometers can be calibrated for accuracy just like other thermometers
In calibration labs like the in-house UKAS certificated ETI Calibration Laboratory at our factory in Worthing, our technicians use industrial black bodies to calibrate infrared thermometers. You can contact them by emailing service@etiltd.com or ask for the calibration lab on 01903 202151
If neither an industrial black body or a comparator cup are available, however, you can do a quick calibration using a properly made ice bath.
Food borne bacteria usually land on the surface of foods first, so infrared thermometers can be useful for spot checking the holding temperatures of plated foods, serving areas, buffets and warming trays.
But infrared thermometers only measure surface temperatures, so they are not very effective at measuring how well cooked food is. Use traditional probe thermometers for this. Critical food safety temperatures, like 5°C and 60°C, should always be verified with an internal probe.
Fortunately there are three ETI infrared thermometers ideal for this. The RayTemp 8 and RayTemp 38 come with attachable type K probes, whilst The Thermapen IR thermometer has an integral foldaway probe to enable internal temperature measurements to be taken.
Infrared thermometers only measure surface temperatures, so they are not very effective at testing if food is properly cooked. Use traditional probe thermometers for this.
If using an infrared thermometer with liquids like soups and sauces, ensure you stir vigorously before taking a measurement to more closely approximate the internal temperature of the liquid. Be aware that steam, even when a liquid is not boiling, can condense on your thermometer and affect the accuracy of your measurements.
Aiming an infrared thermometer at a porous surface like a grill or grate, will factor in the surface temperature of whatever surfaces are visible through the holes of the grill or grate when calculating a final temperature for your reading.
To accurately measure the temperature of a porous grate or grill, place a solid surface like an iron plate or skillet on the grill, let it come to temperature and measure the plate or skillet. Then spray a little cooking oil on the plate or skillet to ensure proper emissivity.
Our range of infrared thermometers require different batteries and have varied battery lives.
Below is a list for each:
1. Mini RayTemp infrared thermometer- Battery type: 9-volt PP3 alkaline. Battery life: 80 hours continuous use.
2. IR-Pocket Thermometer – Battery type: 2 x 1.5-volt AAA. Battery life: 20 hours continuous use.
3. RayTemp 2 high accuracy Infrared Thermometer- Battery type: 3 x 1.5-volt AAA. Battery life: 5000 hours continuous use. RayTemp 2 Plus infrared thermometer with automatic 360° rotating display – Battery type: 3 x 1.5-volt AAA. Battery life: 5000 hours continuous.
5. RayTemp Blue Infrared Thermometer – Battery type: 3 x 1.5-volt AAA. Battery life: 3000 hours.
6. RayTemp HSE IR Thermometer – Battery type: 3 x 1.5-volt AAA. Battery life: 100 hours with backlight, 300 hours without backlight.
7. RayTemp 3 Infrared Thermometer – Battery type: 2 x 1.5-volt AAA. Battery life: 140 hours continuous use.
8. RayTemp 8 infrared thermometer with type K thermocouple socket, RayTemp 38 infrared thermometer & Infra-red thermometer kit – Battery type: 2 x AAA alkaline. Battery life: 180 hours continuous use.
9. Thermapen IR infrared thermometer & Thermapen IR with air probe – Battery type: 2 x 3-volt CR2032 lithium coin cell – Battery life: 1000 hours continuous use.
10. No touch Forehead Thermometer – Battery type: 2 x AAA Alkaline (Supplied). Battery life: 3000 hours (auto-off after 60 seconds).
The "spot size" of any measurement is controlled by two variables:
1. The "distance to target ratio" or "spot ratio" of your infrared thermometer.
2. The distance between your infrared thermometer and the target.
Usually listed on the thermometer itself, the "distance to target ratio" (DTR) or "spot ratio" tells you the diameter of the "circle" of surface area an infrared thermometer will measure at a given distance.
For example, an infrared thermometer with a 12:1 ratio will measure the temperature of a 1" diameter circle of surface area from 12" away, a 2" diameter circle of surface area from 24" away, and so on.
"Emissivity" is a measure of a material's relative ability to emit radiated energy. It is measured on a scale from just above 0.00 to just below 1.00.
Emissivity depends on factors such as temperature, emission angle, and wavelength. Generally, the closer a material's emissivity rating is to 1.00, the more that material tends to absorb reflected or ambient infrared energy and emit only its own infrared radiation. Most organic materials, including the byproducts of plants and animals, have an emissivity rating of 0.95.
Check your infrared thermometer to see if it has adjustable emissivity settings as a feature. Then check your target material against this Emissivity Table.
Use a patch to get accurate measurement. Another way to get an accurate temperature measurement on a material with a low emissivity rating is to cover it with something with a high emissivity rating and let it come to temperature.
For example, a polished metal skillet can be covered with a thin layer of cooking oil, which has an emissivity rating of 0.95. Be sure to allow time for the cooking oil to come to temperature before taking your measurement. But once they are the same temperature, the highly emissive cooking oil makes checking the skillet temperature easy.
The temperature of other metals can be measured more accurately by spraying a spot with flat black paint or by applying a few pieces of black electrical tape and allowing it to come to temperature - both have an emissivity rating of 0.95. When using this method, however, be very careful that the field of view for your measurement does not extend beyond the blackened spot or your reading will be skewed by the surrounding metal.
This is also a great way to get a reading on a non-organic surface with an infrared thermometer that has fixed emissivity.
Fixed emissivity is a setting on some infrared thermometers, usually of 0.95 or 0.97, that attempts to simplify their operation while leaving them suitable for most material surfaces, including almost all foods. Other infrared thermometers come with adjustable emissivity settings, so you can more accurately prepare your infrared thermometers for the type of surface being measured, particularly when measuring non-organic surfaces.
ETI infrared thermometers with Fixed Emissivity:
Mini Raytemp Infrared Thermometer
ETI infrared thermometers with adjustable emissivity:
Highly-polished metals typically have very low emissivity ratings, as they tend to be very reflective of ambient infrared energy and less effective at emitting their own electromagnetic waves. If you point an infrared thermometer with fixed emissivity at a stainless steel pot filled with boiling water, you might get a reading closer to 100°F (38°C) than 212°F (100°C). That's because the shiny metal is better at reflecting the ambient radiation of the room than it is at emitting its own infrared radiation.
Some infrared thermometers have fixed emissivity settings of (usually of 0.95 or 0.97) to simplify their operation while leaving them suitable for most material surfaces, including almost all foods.
Other infrared thermometers come with adjustable emissivity settings, so you can more accurately prepare your thermometer for the type of surface being measured, particularly when measuring non-organic surfaces.
An Infrared thermometer will NOT measure temperature accurately through glass, liquids or other transparent surfaces even though visible light (like a laser) passes through them. If you point an infrared thermometer at a subject through a closed window, you will be measuring the surface temperature of the window itself, not the object you are aiming at.
No. As in the answer above and for the same reasons, an infrared thermometer will only measure the surface temperature of water, not the object's temperature.
If using an infrared thermometer with liquids like soups and sauces, pull a ladle full of liquid up from the bottom of the pot before taking a measurement to more closely approximate the internal temperature. To measure semi-solids like stuffing, corn or mashed potatoes, insert a spoon into the center of the material, pulling it back to create a void and point your infrared thermometer into the void.
Infrared thermometers used to measure temperatures inside a refrigerator or freezer unit should typically be stored inside the fridge or freezer so they are ready to start taking measurements when you need them. To measure the contents of frozen pallets, be sure to break open the pallet, remove at least one box and point your thermometer at the top of one of the center boxes to be sure your reading reflects the temperature of the frozen material inside the pallet and not just the surfaces that are exposed to warmer air.
Thermapen FAQ
The Thermapen is the fastest, most accurate and sensitive thermometer on the market. Our products are hand-manufactured at our factories in Worthing, West Sussex. Each Thermapen is supplied with a certificate of calibration, certifying that it was hand-calibrated in a laboratory against a precision thermometer and found to be accurate at 0 °C (ice point) and 100 °C (boiling point).
This certificate of calibration is traceable, via international agreement, to all major National Standards including UKAS and NIST.
The incredible speed of our Thermapen thermometers is a result of high-quality equipment and rigorous manufacturing processes. The closer the thermocouple wire is to the base of the probe, the faster the response. We thread the wires in by hand and conduct speed tests afterwards to check they meet their specification. Any probes which fall outside of range have to be completely redone.
Most of our digital thermometers will be affected by the emf (electro-magnetic field) generated by the hob and there have been various studies as to the inherent dangers to humans in close vicinity to the active surfaces.
Manufacturers recommend that metal spoons are not to be used, we are concerned that any metal in the design of a probe will be affected, and therefore produce errors.
At this time, we would recommend recording results by heating pans, removing them from the influence of the hob and quickly taking a test measurement. We appreciated that rapid cooling will reduce temperature results, we also suggest an infrared thermometer like The IR-Pocket thermometer (814-060) which has adjustable emissivity or perhaps a Mineral Insulated probe.
Although the Thermapen ONE & Professional are waterproof and can survive short-term submersion, they cannot be put in the dishwasher. The safest, most hygienic way to clean the body of your Thermapen is using anti-bacterial wipes or spray cleaner.
The temperature of food is very dynamic, particularly while it is cooking, and the Thermapen is fast and accurate enough to show minute changes in the temperature while they occur.
The probe tip of the Thermapen can be pushed through food helping you find the coolest part of the centre and can show differences in temperature from one spot to another (different parts of a whole turkey can differ by as much as 11°C).
While it can take seconds for the Thermapen to move from the ambient temperature (room temperature) to the temperature of a food or liquid, once its micro-thermocouple has honed in on the current temperature, it is very quick to sense and display changes (the display refreshes every half-second).
Sometimes people have a mentality that cooking thermometers should be like bathroom scales or speed guns and lock in on the temperature once it is detected, but rest assured, if the Thermapen numbers are changing, the food’s temperature is changing!
Of course, these minor changes can be hidden by changing the display resolution of the Thermapen from tenths to whole numbers (see our How To videos for customising your Thermapen).
You can personalise your Thermapen by changing the factory default settings, including:
Changing the display units from °C to °F,
changing the display resolution from 0.1° to 1° and disabling the auto-off feature.
To see how to do that, please refer back to the operating instructions under the product listing tab Downloads. Or click on the product you wish to personalise below:
We have a range of Thermapens and some require different batteries and have varied battery lives. Below is a list for each:
1. Thermapen Professional Thermometer – Battery type: 1.5-volt AAA. Battery life: 3,000 hours.
2. Thermapen Surface, Thermapen Air, Thermapen Classic, Thermapen Classic with strong probe, Thermapen Sous Vide thermometer & Thermapen Burger probe - Battery Type: 2 x 3-volt CR2032 lithium coin cell. Battery life: 1,500 hours.
3. Thermapen Sous Vide thermometer, & Reference Thermapen – Battery type: 2 x 3-volt CR2032 lithium coin cell. Battery life: 1000 hours continuous use.
4. Thermapen ONE- Battery type: 1.5-volt AAA. Battery life: 2000 hours (without backlight), 100 hours backlight on time
5. Thermapen Blue wireless thermometer- Battery type: 1.5 volt AAA. Battery life: 1,000 hours.
• Thermapen ONE - 1 x AAA
Please see the video which demonstrates clearly how you can change the battery on your Thermapen ONE.
A low battery symbol will appear when the battery needs replacing, in this condition the backlight is set to a low level to save battery life. The instrument continues to measure accurately but we recommend that the battery is changed as soon as possible.
To replace the battery, loosen the battery cover screw using a Pozi (PZ1) screwdriver to remove the battery cover. Replace with a single AAA battery noting the polarity. Tighten the battery cover screw until the battery cover is snug onto the seal. Do not over-tighten.
If you are having any issues replacing the batteries, just contact us via our online contact form and a member of our dedicated team will be in touch to assist.
• Thermapen Classic - 2 x 3 volt CR2032 lithium coin cell
Please see the video which demonstrates clearly how you can change the battery on your Thermapen Classic.
A battery symbol indicates that the batteries will need replacing. The instrument continues to measure accurately but we recommend that the batteries be changed as soon as possible.
To replace the batteries, remove the battery cover using a coin. Remove the batteries by pulling the battery retaining clip back whilst holding the unit upside down. Replace both batteries, positive side up, replace cover.
If you are having any issues replacing the batteries, just contact us via our online contact form and a member of our dedicated team will be in touch to assist.
• Thermapen Professional - 1 x AAA
Please see the video which demonstrates clearly how you can change the battery on your Thermapen Professional.
A low battery symbol will appear when the battery needs replacing, in this condition the backlight is set to a low level to save battery life.
The instrument continues to measure accurately but we recommend that the battery is changed as soon as possible.
To replace the battery, remove the battery cover with a pozi (PZ1) screwdriver. Remove the battery by pulling the battery retaining clip back (do not over extend). Replace the battery, positive end first before screwing the battery cover back down.
Please note: Do not use excessive force when refitting the battery cover ensuring it is compressed against the seal.
If you are having any issues replacing the batteries, just contact us via our online contact form and a member of our dedicated team will be in touch to assist.
Cheaper thermometers are limited by thetechnology that they use. These can be mass produced for just a few pennies to a few pounds. On the other hand, the Thermapen is hand-assembled and hand-tested in the UK and uses a professional thermocouple circuit design. This design and quality cost more to make than cheaper, less capable thermometers.
Thermometer Probe FAQ
ETI makes Thermocouple, Themistor, PT 100/RTD and bespoke probes. For any special requests take a look at our Bespoke Probes page.
• Only use the probe for liquids and semi-solid foods.
• Do not 'stab' food, careful insertion will penetrate most food.
• Do not lift heavy food items with the probe tip.
• Do not strike bones with the probe tip.
• Do not use the probe as an ice pick.
• The probe shaft and tip should not be bent.
• For frozen foods place the probe tip between two frozen packs or use a drill to make a hole in the food before placing the probe tip in the hole.
• Do not create a hole with the probe tip.
• Do not use the probe to pry or puncture non-food items.
• Do not expose the probe tip to flames or temperatures beyond the probe temperature limit.
• Do not expose the handle to high temperatures.
• Do not coil the cable tightly around the handle.
• Avoid excess heat that may melt the cable.
• Avoid excess strain, crimping or stretching of the cable.
• Do not lift the instrument by the probe or cable.
• Keep the lead connector and instrument clean and dry.
• When plugging the probe into the unit, avoid bending pins.
• Do not immerse the probe handle in any liquid (except waterproof probes).
• Damage is not covered by the probe warranty.
Use probe wipes to clean the probe after each food measurement to avoid cross-contamination, wiping both the handle and cable.
Other food-safe cleaning solutions may be used with paper towels in place of probe wipes.
The temperature of food is very dynamic, particularly while it is cooking. For hot foods, find the coldest part of the food by placing the probe tip at the thickest part of the food. For cold foods, find the warmest part of the food. For the most accurate measurements avoid the sides of any container.
Sometimes people have a mentality that cooking thermometers should be like bathroom scales or speed guns and lock in on the temperature once it is detected, but rest assured, if the numbers are changing, the food’s temperature is still changing.
Temperature probes are precision measurement devices. At the probe tip there is an electronic sensor connected by wires running through the probe tube. With proper care, this probe will last a long time, providing accurate measurements.
• Do not 'stab' food, careful insertion will penetrate most food
• Avoid striking bones with the probe tip
• The probe shaft and tip should not be bent
• Do not lift heavy food items with the probe tip
• Do not use the probe as an ice pick
• For frozen foods place the probe tip between two frozen packs or use a drill to make a hole in the food before placing the probe tip in the hole. Do not create a hole with the probe tip
• Do not use the probe to pry or puncture non-food items
• Do not expose the probe tip to flames or temperatures beyond the probe temperature limit
• Do not expose the handle to high temperatures
• Do not coil the cable tightly around the handle
• Avoid excess heat that may melt the cable
• Avoid excess strain, crimping or stretching of the cable
• Do not lift the instrument by the probe or cable
• Keep the lead connector and instrument clean and dry
• When plugging the probe into the unit, avoid bending pins
• Do not immerse the probe handle in any liquid (except waterproof probes)
• Damage is not covered by the probe warranty
Data Logger FAQ
Each ETI data logger comes with its own software. You can download the software when you visit our dedicated Software & Apps page.
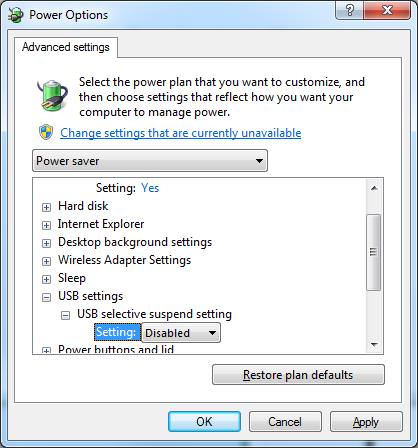
This may be because of the feature of Windows, USB Selective Suspend which activates and turn the USB port power off to the base station.
1. Open the power settings for your computer.
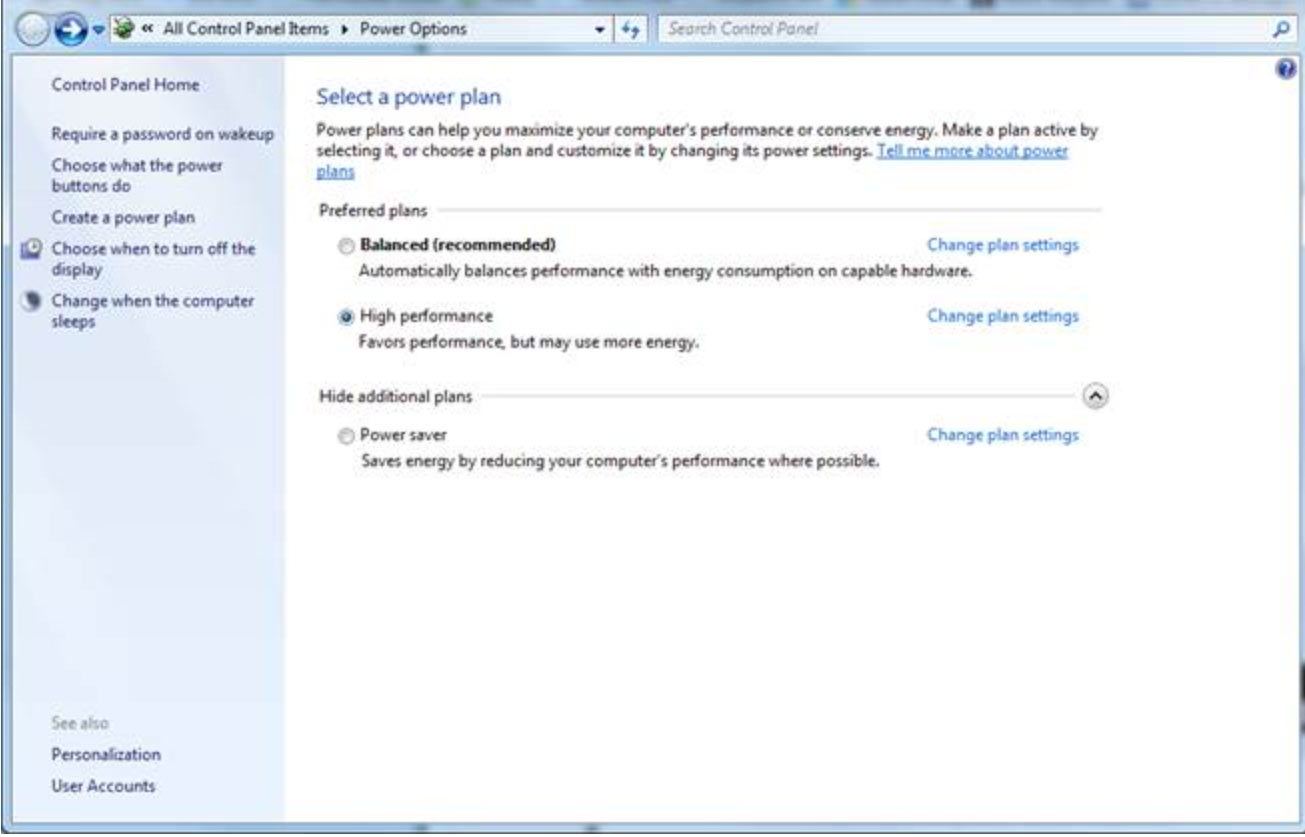
2. Click "change plan settings".
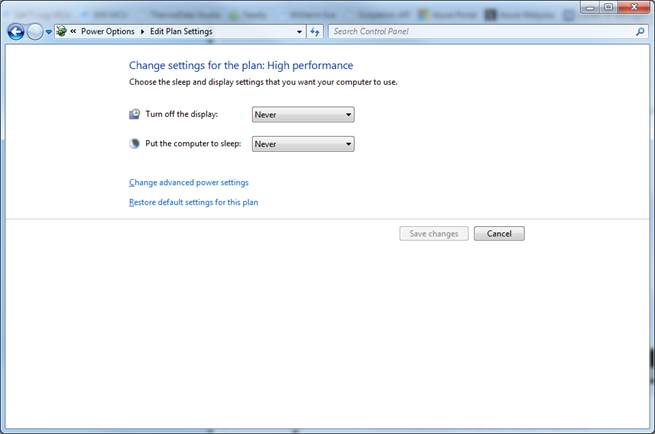
3. Choose"Change Advanced Power Settings".
ETI care for the environment. Therefore all our logger systems don't come with a resource wasteful, printed copy of the user manual, instead it is included on the software installation CD that came with your datalogger. It may also be downloaded directly from the individual product pages on this website by visiting the appropriate logger page and then clicking the Download tab.
One of the key settings for your logger units is to give each different unit a name to differentiate it from the other units on the same network. Most people use either a number or a location or type of material being measured to name their loggers (i.e. "logger1" or "upper_freezer" or "raw_fish").
Some of our data loggers come with built-in cable connectors or wireless capability that can connect directly with your computer, others require the purchase of a separate cradle or data dock that connects directly with your computer through your USB port.
The most common problem in getting your logger up and running is loading the correct drivers. This can usually be ascertained by looking in the System Device Manager on your PC.
Another common problem is failure to install the required software and registerering each logger unit before installing them. If you need help, please don't hesitate to contact our Service Department aftersales@etiltd.com, 01903-202151.
Yes. Some of our data loggers come with preset defaults and are ready to use right out of the box but others will only work once they've been set up by selecting settings directly on the logger unit or by using a computer-driven wizard and registering each unit with the controlling software. Even units with preset defaults should be customized to your particular needs (high and low alarms, frequency of measurements, etc.) before use.
Manometer FAQ
A manometer, otherwise known as a pressure meter or pressure gauge, is a portable device that measures the pressure of a gas or liquid in a system.
You can buy analogue or digital manometers, though digital options are often preferred due to their flexibility, reliability and wide range of units. Manometers often measure either absolute, gauge or differential pressure or a combination of the three.
Manometers are most commonly used by gas HVAC professionals to identify issues such as leaks in heating system pipes. Some specific applications include:
• flue draught measurement
• gas pressure in heating appliances
• air conditioning ducts
• monitoring of filter differential pressure
• velocity measurement with a pitot tube
Digital manometers usually work by connecting the device to the system you’re measuring using a hose. Simply program the device to display readings in your desired unit, connect the hose and carry out the required test. The manometer screen will display the pressure in your chosen unit.
It’s important when taking pressure measurements to be clear about which measurement to use: absolute, gauge or differential. Each is appropriate for different applications.
Absolute pressure
Vessels that contain no molecules, matter or atmospheric pressure, like a vacuum, have zero pressure. Absolute pressure is when pressure is measured on a scale that uses this zero as its reference point.
Absolute pressure readings are best for applications where they are not influenced by changes in atmospheric pressure, like completely sealed, non-flexible containers.
Gauge pressure
Gauge pressure uses the current atmospheric pressure as its reference point instead of zero. A gauge pressure reading is an absolute pressure reading minus the atmospheric pressure of the space/container you are measuring.
Gauge pressure readings are best for applications that are influenced by atmospheric pressure changes, such as an open container.
Differential pressure
Differential pressure is used when the reading and reference values are variable. It is calculated by subtracting one of these values from the other. For example, if measuring the flow of gas along a pipeline and the pressure is 20 psi at one end and 50 psi at the other, the differential pressure is 30 psi.
Differential pressure readings are best for applications where users need to know the pressure difference between two places.
pH Instrumentation FAQ
pH meters can be used in many areas, for example:
Food production
Water quality
Process industry
Laboratories
Fish farming
Aquariums
Agriculture
pH meters work by measuring minute particles, called ions, with two glass electrodes, one of which has a permeable junction. This permeable junction allows a small amount of gel solution to pass through to complete a liquid bridge. If this junction is allowed to dry out, the liquid bridge is prevented from being formed and the instrument will not perform accurately. Therefore, unlike most instruments, the glass electrodes need to be kept wet with a storage fluid for the junction not to get blocked.
The pH meter has a finite life due to the gradual aging of the sensor and will only last for approximately 360 readings (or a year for one reading a day).
If you do not look after your pH meter then incorrect measurements of pH levels may occur. As a minimum, you must always wash the pH electrode (the measuring part of the instrument) in clean water. Do not touch the pH electrode unless using a damp tissue or cloth for cleaning and then only using extreme care.
When not in use, ensure the pH meter electrode is kept moist in either storage solution or a pH 4 solution. If the sensor is allowed to dry out completely, the instrument’s performance will be effected and its warranty invalidated.
If an electrode has been allowed to dry out or becomes slow to respond, it may be rejuvenated by soaking the electrode overnight in storage solution. After overnight soaking, rinse the electrode and then soak in a 4 pH buffer solution before giving the electrode a final rinse. The electrode should then be ready for use.
Depending on the accuracy required, once a day if several measurements are required during the day; or at least once weekly if you use 2 or 3 times per week.
To ensure accurate measurements, it is necessary to calibrate pH meters on a regular basis. For this you will require pH buffer solutions. These standard, inexpensive solutions are used to check that the pH reading is correct.
If it is not, it can be easily corrected following the procedure for the specific instrument. Generally pH electrodes have a limited working life, dependant on the frequency of use.
Electrode was dry or not properly conditioned. Reference junction is possibly clogged up. Remedy: Pre-soak the electrode for 1 hour in tap water before using.
All pH glass electrode will age with time and temperature. Most glass electrodes have a storage life of about 1 year. A life-span of about 3 to 6 months for frequent/continuous use.
Temperature is important as liquid properties are affected. Therefore, a correct temperature of 25 °C for Buffer Solutions should be maintained during calibration. However correction of values may be made if the temperature of the solution is known, by comparing with the table below. Different temperatures of test samples will give inconsistent results.
When the electrode is held in the air and not dipped or in contact with a solution, it will display erratic readings. This is because the reference and glass electrode are in close contact, therefore the electronics cannot show a fixed reading. Dip the electrode into any solution and a reading will appear.

